
(in other words we reduced 100% to decimal form 1. We will let 6Li = x and 7 Li = 1-x we use 1 – x instead of 100 – x because the small number is easier to work with. Since I don’t know what the percentage are, I will have to use variables.ġ00% of Lithium is determined by these two naturally occurring isotopes. Determine the percent abundance of each isotope.Īw = + + Ħ.94 = + The atomic mass of lithium is 6.94, the naturally occurring isotopes are 6Li = 6.015121 amu, and 7Li = 7.016003 amu. Atomic mass for Cu = 63.546Ħ3.546 = + Ħ5Cu = 1 – x = 1 – 0.6916 = 0.3084 x 100% = 30.84% The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z). The mass of 1 mol of bilirubin is 584.73 g. Multiplying the molar mass of each atom by the number of atoms of that type in bilirubin’s formula and adding the results, we get. What are the percent abundances of the isotopes? Since the overall atomic weight for copper is not given in the problem, you must look it up in the periodic table to work this solution. Summing the molar masses of the atoms in the NaCl formula unit gives. 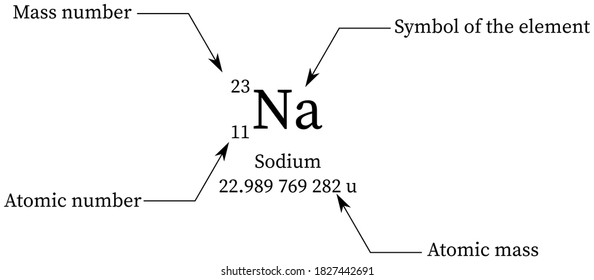
If you look in the periodic table you will be able to check that our answer is correct!ģVerify that the atomic mass of magnesium is 24.31, given the followingĪtomic mass= + + ĭetermining the percent abundance of each isotope from atomic mass.Ĭopper exists as two isotopes: 63Cu (62.9298 amu) and 65Cu (64.9278 amu). Average masses are generally expressed in unified atomic mass units (u), where 1 u is equal to exactly one-twelfth the mass of a neutral atom of carbon-12. Thus, Avogadros number has been measured to be 6.022141×10²³. The average atomic mass (sometimes called atomic weight) of an element is the weighted average mass of the atoms in a naturally occurring sample of the element. 
Or, a little easier to understand, Avogadros number is the ratio of 1 gram to 1 atomic mass unit but with the atomic mass unit expressed in grams. 10.81amu so, the atomic weight of B = 10.81amu Avogadros number is the ratio of the mass of 12 grams of carbon-12 to the mass of 1 atom of carbon-12 measured in grams.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
